Prof. Dr. Andreas Koeberle is a distinguished researcher specializing in the interface of inflammation, cancer, lipid metabolism, and signaling, focusing on uncovering the molecular mechanisms of natural products.
Since 2019, he has been the Head of the Michael Popp Institute at the University of Innsbruck, Austria, where he also holds the position of University Professor for new phyto entities.
He recently obtained a full professorship at the University of Graz in the Department of Pharmaceutical Sciences.
Prof. Koeberle has an extensive publication record, contributing significantly to biochemistry and pharmacology.
His research aims to develop novel therapeutic strategies by exploring the bioactivity of natural compounds, particularly in the context of inflammatory diseases and cancer.
We were grateful for Andreas’ insightful presentation on the link between the membrane lipidome and ferroptosis signaling. Thank you for coming to Vienna, Andreas!

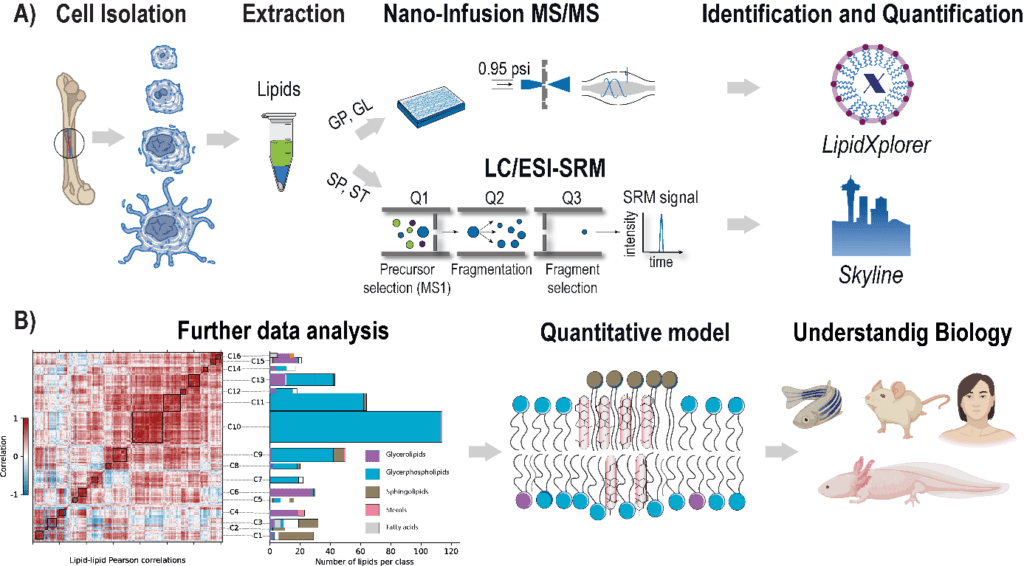







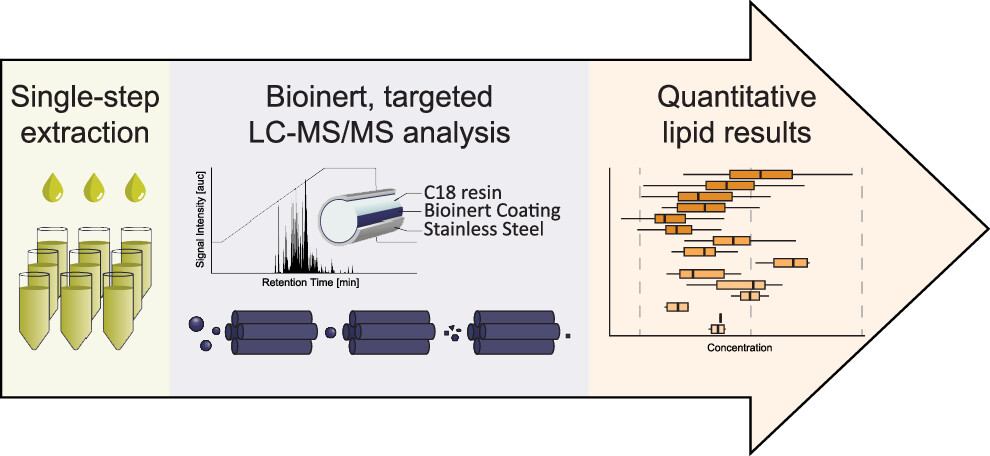

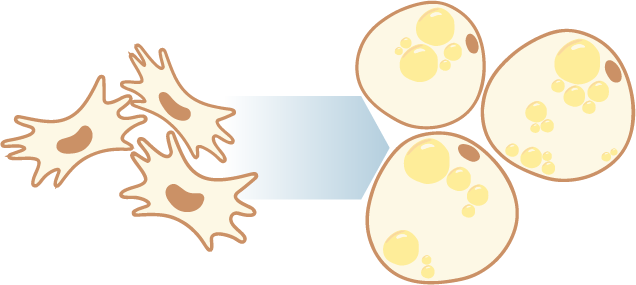 The rising prevalence of obesity globally presents a critical social challenge, threatening to reverse the progress made in life expectancy in developed nations. Gaining a thorough understanding of the mechanisms behind fat cell production is, thus, essential for the development of novel treatment strategies. While past studies focused on individual molecular layers, we recognize the importance of considering higher network connectivity levels, particularly lipid feedback controlling the master regulator of adipogenesis, PPARG. Our approach integrates advanced lipidomics and proteomics techniques, monitoring PPARG and the lipidome during perturbations in vitro. Through innovative multiomics strategies and high-throughput imaging, we dissect feedback networks and identify lipid regulators of PPARG. This research not only advances scientific knowledge but also holds promise for future obesity drug development, potentially revolutionizing treatment approaches.
The rising prevalence of obesity globally presents a critical social challenge, threatening to reverse the progress made in life expectancy in developed nations. Gaining a thorough understanding of the mechanisms behind fat cell production is, thus, essential for the development of novel treatment strategies. While past studies focused on individual molecular layers, we recognize the importance of considering higher network connectivity levels, particularly lipid feedback controlling the master regulator of adipogenesis, PPARG. Our approach integrates advanced lipidomics and proteomics techniques, monitoring PPARG and the lipidome during perturbations in vitro. Through innovative multiomics strategies and high-throughput imaging, we dissect feedback networks and identify lipid regulators of PPARG. This research not only advances scientific knowledge but also holds promise for future obesity drug development, potentially revolutionizing treatment approaches. 





